Polyvinyl chloride is produced by polymerizationof the vinyl chloridemonomer vcm as shown.
Explain the polymerization of vinyl chloride.
Pvc vinyl chloride is an organohalogen compound that has important industrial applications.
For efficient cationic polymerization of vinyl monomers it is necessary that the carbon carbon double bond be the strongest nucleophile in the molecule.
Polymerization of vinyl chloride vc was studied.
About 80 of production involves suspension polymerization.
Vc vc α d 1 vc β β d 2 and vc d 3 were used to study the reactivities of the hydrogen atoms in the polymerization and the β hydrogen atoms contributed to the chain transfer.
The surfactant molecules composed of a hydrophilic water attracting and hydrophobic water repelling end form a stabilizing emulsion before polymerization by coating the monomer droplets.
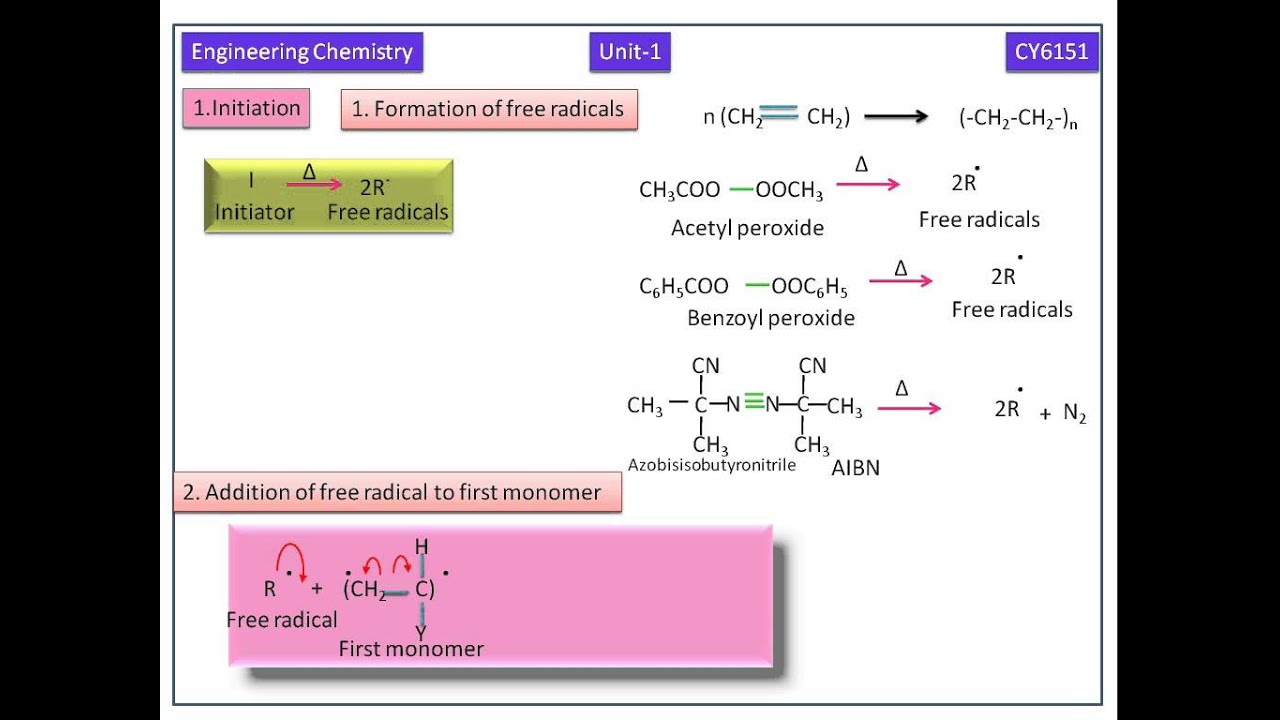
Free radical polymerization frp is a method of polymerization by which a polymer forms by the successive addition of free radical building blocks.
Following its generation the initiating free radical adds nonradical monomer units thereby growing the polymer chain.
Natural evolution of hcl from vc occurred in the polymerization.
Addition polymerization of vinyl compounds or related unsaturated compounds as vinylidene chloride.
Definition of vinyl type polymerization.
Pvc is used in the manufacture of numerous products including packaging films and water pipes.
Polyvinyl chloride is produced by polymerization of the vinyl chloride monomer vcm.
Emulsion polymerizationaccounts for about 12 and bulk polymerizationaccounts for 8.
When treated with certain catalysts vinyl chloride monomers undergo polymerization and form the larger compound known as polyvinyl chloride or pvc.
Other surfactant molecules clump together into smaller aggregates called micelles which also absorb monomer molecules.
The polymerization of vinyl chloride has been studied dilatometrically in chlorobenzene using azoisobutyronitrile as an initiator at temperatures between 30 and 45 c and with monomer and initiator concentrations varying from 1 0 to 8 1 m and from 0 005 to 0 1 m respectively.
A typical polymerization includes 180 parts water and 100 parts vinyl chloride monomer chain transfer agent trichloro ethylene.
The reactants are then heated in the closed system to about 50 c and the pressure rises to about 0 5 mpa.
Thus vinyl acetate would be classed as an electron donor type monomer section 9 10 2 but it cannot be polymerized cationically because the carbonyl group complexes the active center.
About 80 of production involves suspension polymerization.